 Background on Chiral Purity
Background on Chiral Purity
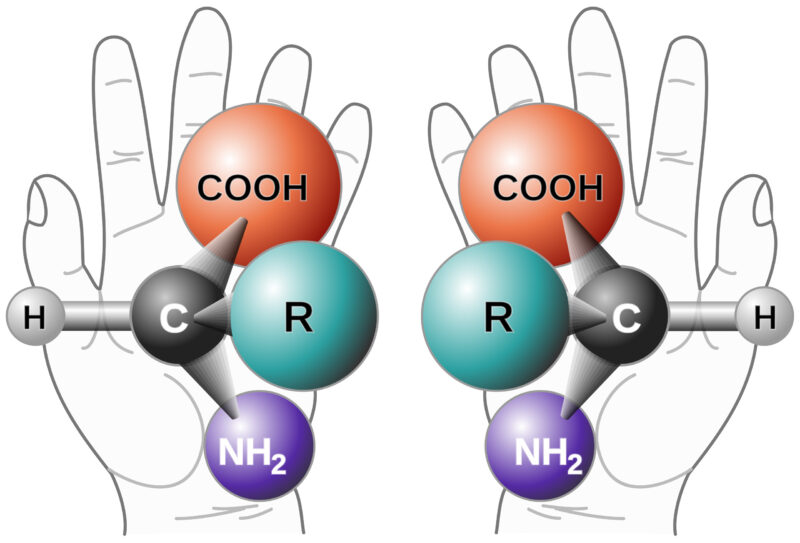
Chirality refers to the phenomenon that occurs when a mirror image cannot be superimposed. It is sometimes called “optical rotation”.
The origin is from the late 19th-century Greek word kheir (‘hand’) and is one of the easiest demonstrations of the concept. Although a person’s hands may appear virtually identical, if they were switched, the outcome would be very different. Amino acids and sugars are the chiral building blocks of larger molecules such as peptides, proteins, and nucleic acids. Therefore, those polymers, in turn, are chiral as well.1 Molecules with chiral centers may have a different therapeutic impact, and this guides the need to test and control chiral purity. The effects of chiral impurities can result in horrific outcomes, as evidenced by the infamous birth defects associated with Thalidomide2 or as benign as Aspartame and sugars (the D/L sugars) that, when superimposed, can create different taste sensations (sweet versus sour, etc.) or metabolic activity. Each chiral center can generate two enantiomers. “Enantiomers or optical isomers are chiral molecules which are non-superimposable mirror images of each other,”3 and multiple chiral centers can generate diastereomers (non-mirror image isomers).
Determining Purity
HPLC has been the primary technique for determining chiral purity, with gas chromatography used occasionally. Measurement of optical rotation is a legacy technique that is fast but not as accurate. Historically, HPLC methods used under normal phase conditions could limit the type of molecules that could be analyzed. However, now modern chiral columns are compatible with reverse phase conditions.
Simulated Moving Bed (SMB) Chromatography as an Option for Chiral Purity Analysis
Do you have a partner for chiral separations? AMPAC Analytical has the expertise in performing chiral purity testing, along with the equipment and techniques. Additionally, our parent company, AMPAC Fine Chemicals, has decades of experience conducting chromatographic separations at a commercial scale in a highly regulated environment. Our services include SMB screening, method development, proof-of-concept demonstration, and production. We operate the largest CGMP Simulated Moving Bed (SMB) chromatography unit in the United States. These technologies and expertise are part of a one-stop shop (from 10-millimeter columns up to 1000mm). Our SMB processes can be developed in a few weeks and are easily scalable. In many cases, scale-up from gram to multi-ton quantities can be achieved in fewer than six months. Our facilities include kilo-scale and pilot-scale units to support smaller quantities, also under CGMP conditions. The SMB facilities have been inspected and approved by the FDA for the manufacturing of APIs. AFC has registered four products with regulatory authorities (FDA/EMA) using SMB technology. Along with chiral separations, we can also perform the separation of diastereomers & regioisomers.
Contact us today for information on how we can assist with your raw material, amino acid, drug product, and API chiral purity testing or to learn more about our SMB processes.
References